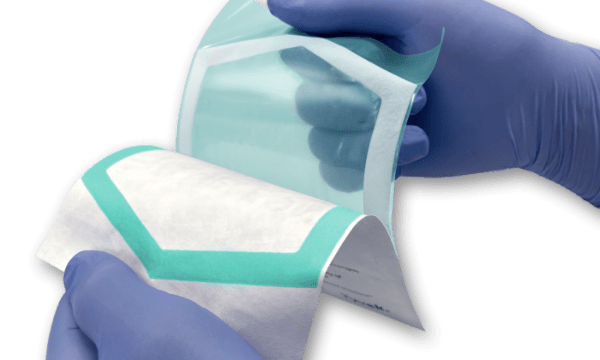
PAXXUS is the leading supplier of engineered flexible materials for the global healthcare market and dedicated to supporting the complex regulatory requirements of the medical device, pharmaceutical, diagnostic, and life sciences industries across the globe.
As a vertically integrated manufacturer, PAXXUS provides tailored material structures to meet your specific packaging requirements. Through constant innovation, PAXXUS has been recognized and awarded for cutting-edge advancements in clear high-barrier films, aluminum foil composites, peelable solutions, and chemically-resistant materials.
In addition to best-in-class material science, our partners can expect agile and responsive support from design to validation and beyond.